| Nakai K et al. | ||||||||
|---|---|---|---|---|---|---|---|---|
|
Boron Neutron Capture Therapy for Glioblastoma: A Phase-I/II Clinical Trial at JRR-4 European Association of NeuroOncology Magazine 2014; 4 (3): 116-123 PDF Summary Figures
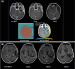
Keywords: adverse event, boron neutron capture therapy, efficacy, glioblastoma, radiation therapy, safety Boron neutron capture therapy (BNCT) is a cell-selective radiation therapy for the treatment of glioblastoma. We reviewed the experience of our BNCT clinical trial and report the present state in 17 cases. Endpoints of our analysis were the efficacy and safety data of BNCT for newly diagnosed glioblastoma. Adverse events seen in our patients included post-treatment brain oedema, convulsion, and diplopia due to swallowing. For newly diagnosed glioblastoma (GBM), median overall survival (OS) reached 25.7 months, and the 1- and 2-year OS rates were 85.7 % and 45.5 %, respectively. BNCT could prolong OS in selected cases. |

Verlag für Medizin und Wirtschaft |
|
||||||||
|
Summary
|
|||||||||
| copyright © 2000–2025 Krause & Pachernegg GmbH | Sitemap | Datenschutz | Impressum | |||||||||