Micardis(R) - Micardis Plus(R) - Verträglichkeit (9 Abbildungen)
| Abbildung |
|---|
| |
|
|
FÜR ÖSTERREICH: Weitere Informationen: Merck Gesellschaft mbH, Zimbagasse 5, 1147 Wien, Tel.: 01/576 00-0
Abbildung 0
|
| |
| |
|
|
Verträglichkeit
Abbildung 1
|
| |
| |
|
|
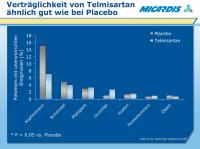
Telmisartan - Placebo - Verträglichkeit
Abbildung 2: Telmisartan has been evaluated for safety in 27 studies in a total of 5363 patients with essential hypertension, including 2921 patients treated for up to 6 months, 888 patients treated between 6 and 12 months, and 1554 patients treated for 1 year or more.1
Adverse events were typically mild and transient in nature.
In placebo-controlled trials that included 1758 patients treated with doses ranging from 20 mg to 160 mg (1344 patients received telmisartan monotherapy, 414 received telmisartan/HCTZ combination), the incidence of adverse events was comparable to that of placebo.1
Headache was the most frequently reported adverse event (7.1% in telmisartan-treated patients vs 15.1% in the placebo group).1
Data on file. Boehringer Ingelheim GmbH.
Keywords: Diagramm,
Telmisartan,
Verträglichkeit
|
| |
| |
|
|
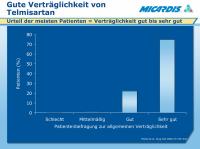
Telmisartan - Verträglichkeit
Abbildung 3: A German post-marketing surveillance study followed 19,870 patients treated with telmisartan for 6 months.1
Data were not available for 2% of patients. Of the rest, 98.8% gave a global rating of tolerability of good or very good.
Adverse events were reported by 1.9% of patients, the most frequent being headache (0.3%), dizziness (0.2%) and nausea (0.2%).
Age, gender and comorbidities had no effect on global tolerability.
1. Michel MC, et al. Safety of telmisartan in patients with arterial hypertension. An open-label observational study. Drug Saf 2004; 27: 335–344.
Keywords: Diagramm,
Patientenbefragung,
Telmisartan
|
| |
| |
|
|
Telmisartan - HCTZ - Verträglichkeit
Abbildung 4: Safety data were collected from 34 multicentre randomized studies, the majority of which were double-blind, which recruited 6,915 patients.1
Patients received placebo, telmisartan 10–160 mg, or telmisartan 10–160 mg + HCTZ 6.25–25 mg for a minimum of 7 days and up to >2 years.
819 patients were treated with placebo, 6575 treated with telmisartan, and 2180 patients were treated with telmisartan + HCTZ.
When calculated according to exposure to study drug, treatment-related adverse events were low with both telmisartan monotherapy and combination therapy.
The incidence of adverse events were similar in younger (< 65 years) and elderly (≥ 65 years) patients.
1. Mancia G. Tolerability and safety of telmisartan as monotherapy or combined with hydrochlorothiazide compared with placebo. Am J Hypertens 2002; 15 (Supp 1): A54.
Keywords: Diagramm,
HCTZ,
Telmisartan,
unerwünschtes Ereignis,
Verträglichkeit
|
| |
| |
|
|
Verträglichkeitsprofil
Abbildung 5
Keywords: Verträglichkeit
|
| |
| |
|
|
Verträglichkeitsprofil
Abbildung 6: In this study, 578 patients with mild-to-moderate hypertension received telmisartan 40–160 mg or lisinopril 10–40 mg once daily for 52 weeks.1
At the end of the maintenance period, mean SBP/ DBP reductions were 23.8/16.6 mmHg and 19.9/15.6 mmHg in the telmisartan and lisinopril arms, respectively.
Treatment-related side effects occurred in fewer telmisartan-treated patients (28%) than in lisinopril-treated patients (40%, P=0.001), principally due to an increase in cough with lisinopril.
There were also two cases of angioedema, both in the lisinopril group.
Similar results were seen in an 8-week comparison of telmisartan with enalapril in 86 patients with severe hypertension.2 No patients on telmisartan experienced cough during this study, compared with 7% of patients on enalapril.
Neutel JM, Frishman WH, Oparil S, et al. Comparison of telmisartan with lisinopril in patients with mild-to-moderate hypertension. Am J Therapeutics 1999; 6: 161–166.
Neutel JM, Smith DHG, Reilly PA. The efficacy and safety of telmisartan compared to enalapril in patients with severe hypertension. Int J Clin Pract 1999; 53: 175–178.
Keywords: Diagramm,
Lisinopril,
Telmisartan,
Verträglichkeit
|
| |
| |
|
|
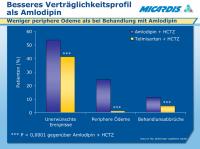
Verträglichkeitsprofil
Abbildung 7: This study, A comparison of Telmisartan plus HCTZ with amlodipine plus HCTZ in Older patients with predominantly Systolic hypertension (ATHOS), compared telmisartan 40–80 mg + HCTZ 12.5 mg and amlodipine 5–10 mg + HCTZ 12.5 mg.1
There were more adverse events in the amlodipine group than in the telmisartan group.
The most frequently reported adverse event was peripheral oedema, reported in 6 (1.2%) patients taking telmisartan + HCTZ, compared with 122 (24.3%) patients taking amlodipine + HCTZ.
This led to a higher overall discontinuation rate from adverse events in the amlodipine group compared with the telmisartan group.
Data on file. Boehringer Ingelheim, GmbH.
Keywords: Amlodipin,
Diagramm,
Telmisartan,
Verträglichkeit,
Ödem
|
| |
| |
|
|
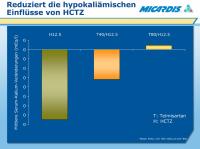
HCTZ - Hypokaliämische Einflüsse
Abbildung 8: Thiazide therapy can cause hypokalaemia, which can, potentially, trigger adverse cardiac events.1,2
Reported here are selected arms from an 8-week, 4 x 5 factorial, multicentre, double-blind, placebo-controlled, dose-finding study conducted in 818 patients with mild-to-moderate hypertension.3
Patients were randomized to telmisartan 20 mg, 40 mg, 80 mg or 160 mg alone or in combination with HCTZ 6.25 mg, 12.5 mg, or 25 mg, giving a total of 20 treatment groups.
HCTZ caused a dose-related decrease in serum potassium.
Combination with telmisartan tended to offset the effects of HCTZ on serum potassium levels.
1. Beermann B, Groschinsky-Grind M. Pharmacokinetics of hydrochlorothiazide in man. Eur J Clin Pharmacol 1977; 12: 297–303.
2. Siscovick D, et al. Diuretic therapy for hypertension and the risk of primary cardiac arrest. N Engl J Med 1994; 330: 1852–1857.
3. McGill JB, Reilly PA. Telmisartan plus hydrochlorothiazide versus telmisartan or hydrochlorothiazide monotherapy in patients with mild to moderate hypertension: a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial. Clin Ther 2001; 23: 833–850.
Keywords: Diagramm,
HCTZ,
Kalium,
Telmisartan
|
| |
| |
|
|
Verträglichkeit
Abbildung 9
|
| |
| |
|