| Abbildung |
|---|
| |
|
|
FÜR ÖSTERREICH: Weitere Informationen: Serono Austria GmbH, Floridsdorfer Hauptstraße 1, 1210 Wien; Tel.: 01/604 7690-0
Abbildung 0
|
| |
| |
|
|
EVIDENCE-Umstellungsstudie
Abbildung 1
|
| |
| |
|
|
EVIDENCE-Studienzentren
Abbildung 2
|
| |
| |
|
|
Patienten - Einschlußkriterien
Abbildung 3
Keywords: Einschlusskriterien
|
| |
| |
|
|
Ausschlußkriterien
Abbildung 4
Keywords: Ausschlusskriterien
|
| |
| |
|
|
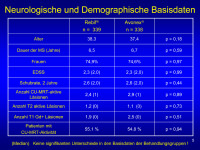
Basisdaten
Abbildung 5
Keywords: Demographie,
Neurologie
|
| |
| |
|
|
EVIDENCE - Chronologie
Abbildung 6
Keywords: Chronologie
|
| |
| |
|
|
Design - Patientenverteilung
Abbildung 7
Keywords: Diagramm,
Patient,
Studiendesign
|
| |
| |
|
|
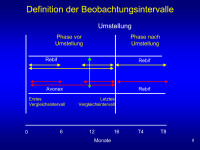
Intervalle
Abbildung 8: EVIDENCE study design:
pre-study MRI scan and clinical assessment;
2 relapses prior 2 years;
randomize at SD1;
monthly MRI gadolinium assessments until week 24;
3-monthly neurological examinations
monthly AE/clinical assessment at time of MRI;
monthly phone check between MRI dates to increase relapse ascertainment and reduce number of historical relapses
reduced frequency of MRI scans after week 24;
primary endpoint at week 24.
Second phase from week 24-48;
Post-48 week is variable because patients enrolled early continue on therapy until last patient enrolled completes week 48.
Transition (T) is when patients switch from Avonex to Rebif.
Keywords: Beobachtung,
Diagramm,
Intervall
|
| |
| |
|
|
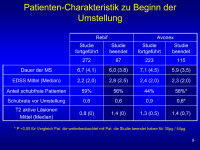
Patientencharakteristik
Abbildung 9
Keywords: Avonex,
Patient,
Rebif
|
| |
| |
|
|
Studienendpunkte
Abbildung 10: The outcome measures for the non-comparative extension phase were changed since there is no validity to compare between original Avonex and Rebif groups due to different Rx regimens prior to T and the fact that dropout reasons are different between groups. Thus the valid comparisons are the within patient comparisons prior to and after conversion to Rebif for the Avonex group and for comparability, the Rebif group pre and post-T.
Similarly the proportion relapse-free cannot readily be compared pre-T and post-T since the time intervals are different and the populations have changed since study start and even comparing the same set of patients is not really valid as patients have been treated for 12-24 months prior to T.
The only measures to assess are relapse and MRI (T2).
Keywords: Studienendpunkt
|
| |
| |
|
|
Umstellung
Abbildung 11
|
| |
| |
|
|
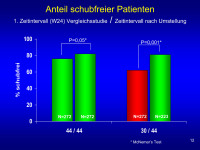
Patienten
Abbildung 12: This slide presents the proportion of patients who remain relapse-free during the post-T interval for both groups and is compared with the original 24 week data from EVIDENCE for the same patients who continue in the post-Transition time.
Slide shows that the difference seen at 0-24 is likely due to Rx since the outcome within each group is considerably different based on dose but between doses is very different in weeks 0 to 24 but very similar once all patients on Rebif
44/44 means Rebif from start and 30/44 is Avonex to Rebif switch group.
Keywords: Patient,
Schubaktivität
|
| |
| |
|
|
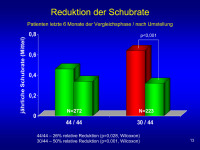
Schubrate
Abbildung 13: Relapse rate providing possibly the most relevant statistical comparison – final interval pre-T and first interval post-T. Both groups improve (?regression over time, fewer contacts, full effect of Rx being seen etc.) but the difference is considerably greater for 30 to 44 than 44/44 and brings the Avonex patient rate to that of Rebif patient rate.
Keywords: Diagramm,
Schubrate
|
| |
| |
|
|
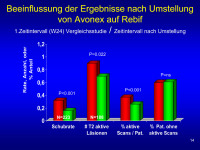
Umstellung Avonex - Rebif
Abbildung 14: 'n’ of 188 reflects number of patients in post transition group (223) who had scans performed – not all patients had scans during EVIDENCE and a number of scans are missing due to errors at site in terms of when scans were to be done.
Y-axis shows either relapse rate, T2 count per patient per scan or the proportion of scans active per patient or proportion of patients with no active scans – values are means (median for T2 lesions and % T2 active scans are both 0).
The ns difference for % with no T2 activity likely reflects 2 issues – based on dose titration, switch patients are still on low dose therapy for first month and any single new lesion renders the patient active for the entire interval. Secondly, the post-T interval is on average 8 months compared to 6 months or less for the pre-T scan.
Otherwise, data shows there is a substantial and immediate effect of switching from 30 qw to 44 tiw.
Keywords: Avonex,
Diagramm,
Rebif
|
| |
| |
|
|
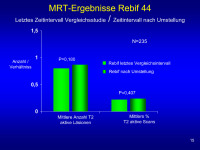
MRT-Ergebnisse - Rebif
Abbildung 15: ‘n’ of 235 reflects number of patients in post transition group (272) who had scans performed – not all patients had scans during EVIDENCE and a number of scans are missing due to errors at site in terms of when scans were to be done.
Y-axis shows either T2 count per patient per scan or the proportion of scans per patient that are active – values are means (median for T2 lesions and % T2 active scans are both 0).
The differences on MRI measures (the more objective measure) are not different before or after T for the 44/44 group. The jump in T2 lesion count is mainly driven by 1 patient who was NAb+ve and who had 8 lesions pre-T and 63 lesions post-T. Removing that single patient makes the mean value for the post-T interval drop to .87
Median values are 0 for T2 active lesions and T2 active scans.
Keywords: Diagramm,
MRT,
Zeit
|
| |
| |
|
|
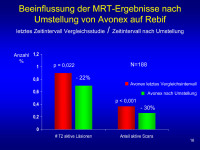
MRT-Ergebnisse - Umstellung - Avonex - Rebif
Abbildung 16: ‘n’ of 188 reflects number of patients in post transition group (223) who had scans performed – not all patients had scans during EVIDENCE and a number of scans are missing due to errors at site in terms of when scans were to be done.
Y-axis shows either T2 count per patient per scan or the proportion of scans active per patient– values are means (median for T2 lesions and % T2 active scans are both 0).
Data shows there is a substantial and immediate effect of switching from 30 qw to 44 tiw.
Keywords: Avonex,
Diagramm,
MRT,
Rebif,
Zeit
|
| |
| |
|
|
EVIDENCE-Umstellung
Abbildung 17
|
| |
| |
|
|
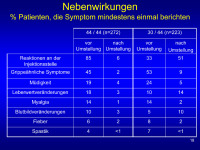
Nebenwirkungen
Abbildung 18: Addresses most common IFN-related adverse events.
Pre-T is pre-Transition rate
Post-T is post-Transition rate
Points to note:
new onset events very uncommon on 44/44 indicating that most patients who develop IFN-related Aes do so early on;
increase in ISR for 30/44 is expected based on change in route of administration;
numbers of new LFT and WBC events is consistent with dose effect of IFN on these events;
number of new FLS is quite low compared to initiation of 30mcg though still greater than new FLS for 44/44 group.
Keywords: Diagramm,
Nebenwirkung
|
| |
| |
|
|
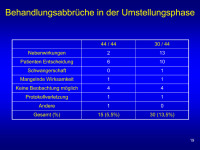
Umstellung - Behandlungsabbrüche
Abbildung 19
|
| |
| |
|
|
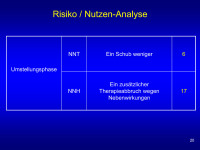
Risiko/Nutzen-Analyse
Abbildung 20
|
| |
| |
|
|
Schlußfolgerungen
Abbildung 21
|
| |
| |
|
|
Schlußfolgerungen
Abbildung 22
|
| |
| |